Another allergen, HDM, has been reported to promote IL-1β secretion in macrophages isolated from asthmatic lungs ( 28). In an OVA-induced asthma model, allergen inhalation induced ROS production, NLRP3 inflammasome components expression and inflammasome assembly in mouse bronchial epithelial cells ( 22, 25– 27). The expression of NLRP3 and IL-18 is also increased in the airway epithelium of asthmatic patients compared with healthy controls ( 24). The expression of NLRP3 and caspase-1 in BAL fluids from asthmatic patients is higher than that in BAL fluids from healthy controls ( 22, 23). Variants in the Nlrp3 gene increase the risk of asthma in patients ( 21).

However, the mechanism by which NLRP3 affects allergic asthma remains unclear ( 20). NLRP3 also promotes the inflammatory response in allergic asthma ( 16– 19). Several inhibitors of the NLRP3 inflammasome have been reported to prevent tissue destruction in NLRP3-related diseases in animal models ( 14) thus, NLRP3 is a potential therapeutic target for these diseases ( 15). The NLRP3 inflammasome can be activated by infections and danger signals released by tissue damage, leading to cell pyroptosis and IL-1β secretion ( 13). Thus, it is important to find new targets in asthma and to provide new treatments with fewer side effects.īy forming the inflammasome complex, NLRP3 functions as a cellular sensor that is involved in many diseases, such as type 2 diabetes, Alzheimer’s disease, inflammatory bowel disease and some infectious diseases ( 11, 12). Side effects, including respiratory tract infections and decreased bone density, often occur with the use of inhaled corticosteroids, which are primary medications in all steps of asthma therapy ( 9, 10). Corticosteroids, β2 agonists and leukotriene receptor antagonists are commonly used in the treatment of asthma ( 8). It has been demonstrated that the recognition of various stimuli and the associated inflammatory responses are important in the development and exacerbation of asthma ( 6, 7). The type II immune response induced by inhaled allergens, infectious agents or air pollution leads to IL-5, IL-4 and IL-13 secretion in the airway, which can lead to eosinophil accumulation, mast cell activation and airway hyperreactivity in allergic asthma ( 3– 5). The airway can come into contact with pathogens, infectious agents, and danger signals present in the environment ( 1, 2). Small molecules targeting the NLRP3 inflammasome may provide new treatment options for this disease. Our results showed that NLRP3 in myeloid cells promoted the development and progression of allergic asthma in an inflammasome-dependent manner. Treatment with RRx-001, an NLRP3 inhibitor, significantly reduced inflammatory cell infiltration and mucus secretion in the airway. Using Nlrp3-mutant mice, we found that NLRP3 promotes the inflammatory response and pathogenesis in HDM-induced allergic asthma in an inflammasome-dependent manner.
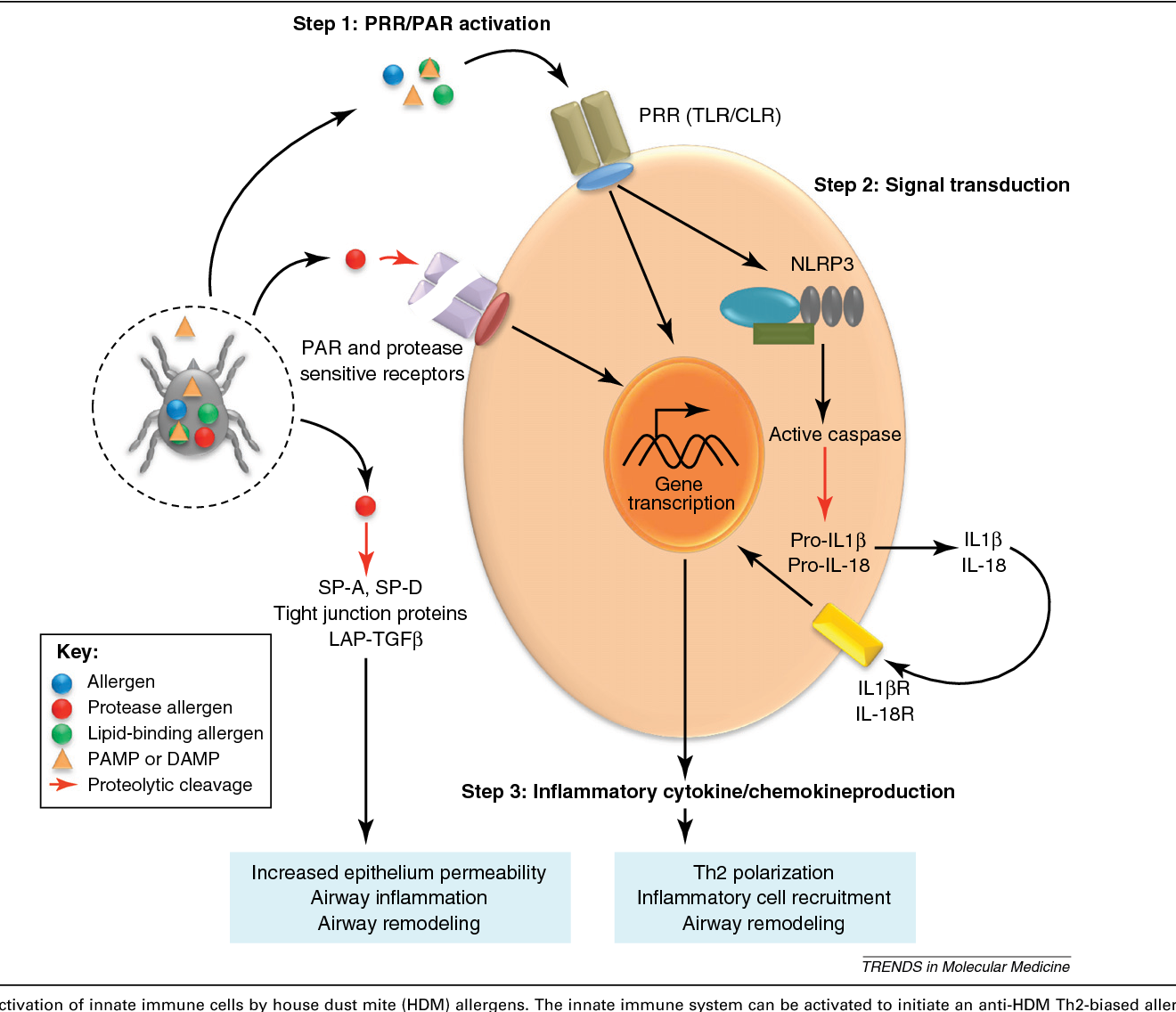
Here, we showed that inhaled house dust mite (HDM) promotes NLRP3 inflammasome activation in the lungs and specifically induces the maturation of caspase-1 and IL-1β in alveolar macrophages (AMs). However, the mechanism by which NLRP3 affects asthma requires further investigation. NLRP3, as an immune sensor of infections and cellular stress, is associated with the development and exacerbation of asthma. Inhaled allergens promote inflammatory response, tissue damage, and airway hyperresponsiveness in the lungs, leading to allergic asthma.
